According to the new market research report " Point of Care Molecular Diagnostics Market by Product & Service (Assays, Kits, Analyzers, Software), Application (Respiratory Disease, Hospital Acquired Infection), Technology (RT-PCR, INAAT), End User (Physician Office, ICUS) - Global Forecast to 2023", published by MarketsandMarkets™,
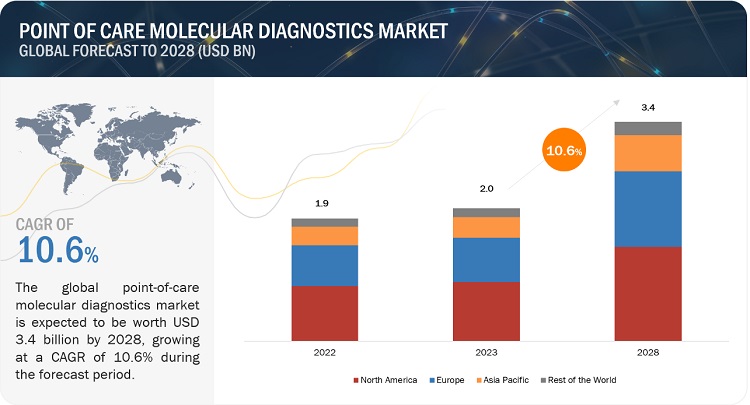
the global point of care (POC) molecular diagnostics market is projected to reach USD 1440.2 million in 2023 from USD 725.5 million in 2018, at CAGR of 14.7%.
Speak to our analyst and gain crucial industry insights that will help your business
POC molecular diagnostics are various point of care portable device and assays & kits used by healthcare professionals to detect and diagnose diseases in human samples such as serum, blood, throat swab, and stool.
In this report, the market has been categorized based on product & service, applications, technology, end user, and region.
Based on the applications, the market is segmented into respiratory diseases, hospital-acquired infections (HAIs), sexually transmitted diseases (STDs), oncology, hepatitis, and other applications (meningococcal meningitis, malaria, Herpes Simplex Virus, Ebola, Zika virus). The respiratory application segment is expected account for the largest share of the global POC molecular diagnostics market in 2018. This large share of this segment can be attributed to the rising prevalence of influenza A & B and increasing research investments by key players for other respiratory disease such as Strep A and TB. In addition to this, most of the FDA approved RT-PCR POC molecular diagnostics are focused on influenza disease.
Browse and in-depth TOC on " Point of Care Molecular Diagnostics Market "
68 - Tables
23 - Figures
106 - Pages
View more detailed TOC @
The POC molecular diagnostics market is broadly segmented into instruments, assays & kits, and services & software based on the basis of product & service. Assays & kits segment is expected to account for the largest share of the global POC molecular diagnostics market and expected to register the highest CAGR during the forecast period. POC molecular diagnostic assays and kits are specially designed for points of care such as hospital critical care units, physician offices, outpatient clinics, and community health posts. POC molecular diagnostics assays and kits enable the early diagnosis of respiratory tract infections, women’s health and sexual health conditions and among others.
The POC molecular diagnostics market is segmented into RT-PCR, INAAT, and others technologies. In 2018, the RT-PCR segment is expected to account for the largest share of the global POC molecular diagnostics market. The large share of this segment can be attributed to the growing use of RT-PCR in proteomics and genomics and access to portable, easy-to-use devices. In the last three years from 2015, more than 42 RT-PCR based products have been launched for various diseases.
In 2018, North America is expected to account for the largest share of the global POC molecular diagnostics market. Growing prevalence of infectious diseases, increasing number of CLIA product approvals, and rising government initiatives are the major factors driving the growth of this market in North America. However, Asia Pacific is expected to grow at highest CAGR during forecast period.
The key players in the global POC molecular diagnostics market are Abbott Laboratories (US), Biomerieux (France), Danaher (US) Roche diagnostics (Switzerland), Meridian biosciences (US), Quidel (US). These players focus on organic strategies such as product launches & approvals, to sustain their growth in the POC molecular diagnostics market.
About MarketsandMarkets™
MarketsandMarkets™ provides quantified B2B research on 30,000 high growth niche opportunities/threats which will impact 70% to 80% of worldwide companies’ revenues. Currently servicing 7500 customers worldwide including 80% of global Fortune 1000 companies as clients. Almost 75,000 top officers across eight industries worldwide approach MarketsandMarkets™ for their painpoints around revenues decisions.
Our 850 fulltime analyst and SMEs at MarketsandMarkets™ are tracking global high growth markets following the "Growth Engagement Model – GEM". The GEM aims at proactive collaboration with the clients to identify new opportunities, identify most important customers, write "Attack, avoid and defend" strategies, identify sources of incremental revenues for both the company and its competitors. MarketsandMarkets™ now coming up with 1,500 MicroQuadrants (Positioning top players across leaders, emerging companies, innovators, strategic players) annually in high growth emerging segments. MarketsandMarkets™ is determined to benefit more than 10,000 companies this year for their revenue planning and help them take their innovations/disruptions early to the market by providing them research ahead of the curve.
MarketsandMarkets’s flagship competitive intelligence and market research platform, "Knowledge Store" connects over 200,000 markets and entire value chains for deeper understanding of the unmet insights along with market sizing and forecasts of niche markets.
Contact:
Mr. Shelly Singh
MarketsandMarkets™ INC.
630 Dundee Road
Suite 430
Northbrook, IL 60062
USA : 1-888-600-6441